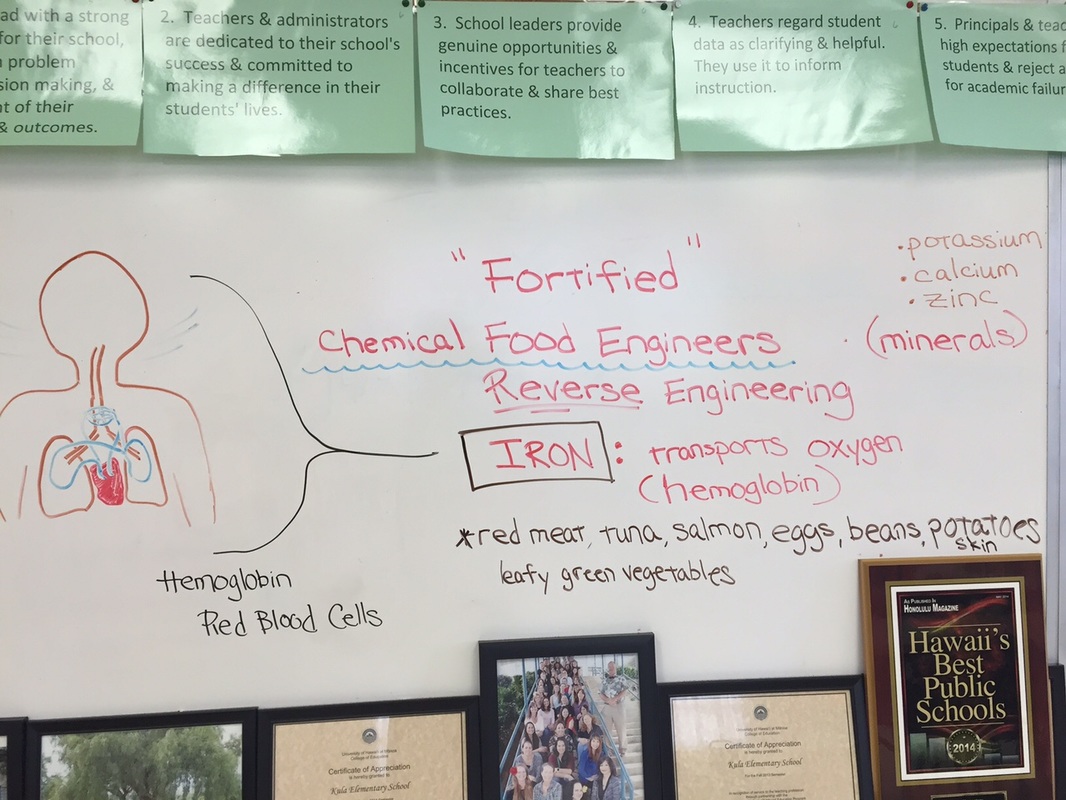
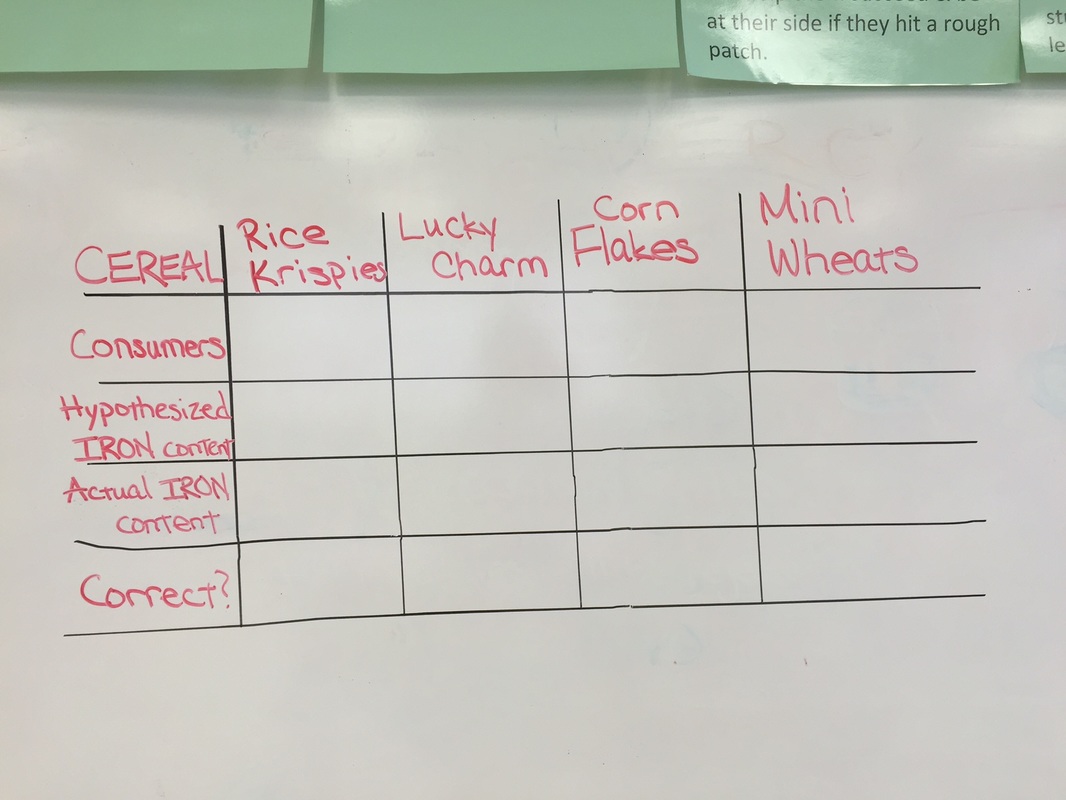
On 10/25, the 2nd and 3rd graders learned one of the roles of a Chemical Food Engineer and we got to experiment with some of our all-time favorite cereals. Chemical Food Engineers often fortify our foods like milk, juice, cereal etc. with additional vitamins and minerals to make sure that we all get our essential nutrients. Their role and their products are especially important when it comes to providing nutrients for people in areas where food is a scarce resource. The human body needs essential minerals like potassium, calcium, zinc, iron and many more to keep us healthy and functioning. For our class today, we are focusing on "iron." Iron is essential in forming hemoglobin which is part of our red blood cells. Hemoglobin transports oxygen in the blood from our lungs to the rest of the body. This is why iron is so important. In our experiment, we attempted to "extract" the iron from 4 popular cereals so that the students can actually see the iron with their naked eyes! To do this, we first had to crush up the cereal in a paper cup and then put it in water so the contents are freely flowing. Then, we used a super strong magnet (your average fridge magnet will not work for this!) and we slowly ran the magnet against the side of the cup and pulled the tiny slate specks of iron up to the surface. This was not an easy experiment but the students persevered and were quite shocked to find these little iron flakes in something they ate every morning. The winning cereal was Frosted Mini Wheats with and iron level of about 90% the daily recommended level!
|
|
AuthorsMrs. Shim and her STRETCH students Archives
May 2015
Categories |



 RSS Feed
RSS Feed
