|
After the Scratch object oriented programming session that led up to the Holiday break, the 2nd graders were ready to come back and get their hands dirty as well as putting their creativity to the test! We did some wonderful projects and explored the science behind bath bombs, fizzy soaps, water molecule attractions (foam art), geometric structure engineering (with gum drops/toothpicks), hot air and tea bag flights, air cannons, how looks affect or even distort your sense of taste (apple juice experiment), polymers (making bouncy balls), tie dye circles, and some physics (in a lady bug race!). This group of students was able to tackle the wide range of projects with a very positive can do attitude. Check them out in action below:
Aloha STRETCH class followers! Back on January 23, 2017, we had our first class of K-1 STRETCH students - very exciting. This is quite a fun and investigative group of youngsters. So far we have explored and created projects on bridges, shaving foam art, building rescue devices, robot hands, water filters, and even an earthquake proof structures. Starting on January 31, we also launched a pilot program - Kindergarten Science Club. These students have been exploring similar experiments and projects. It's been a jam-packed few weeks getting to know these new students and here are some great pictures. Take a look!
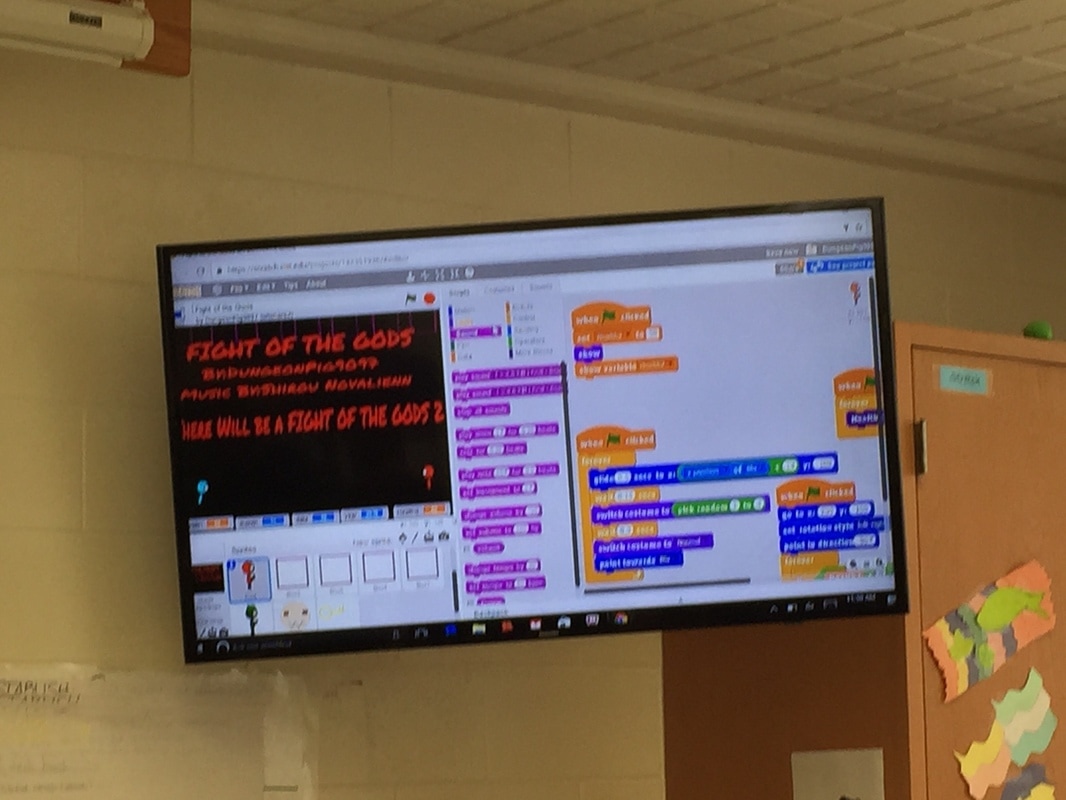
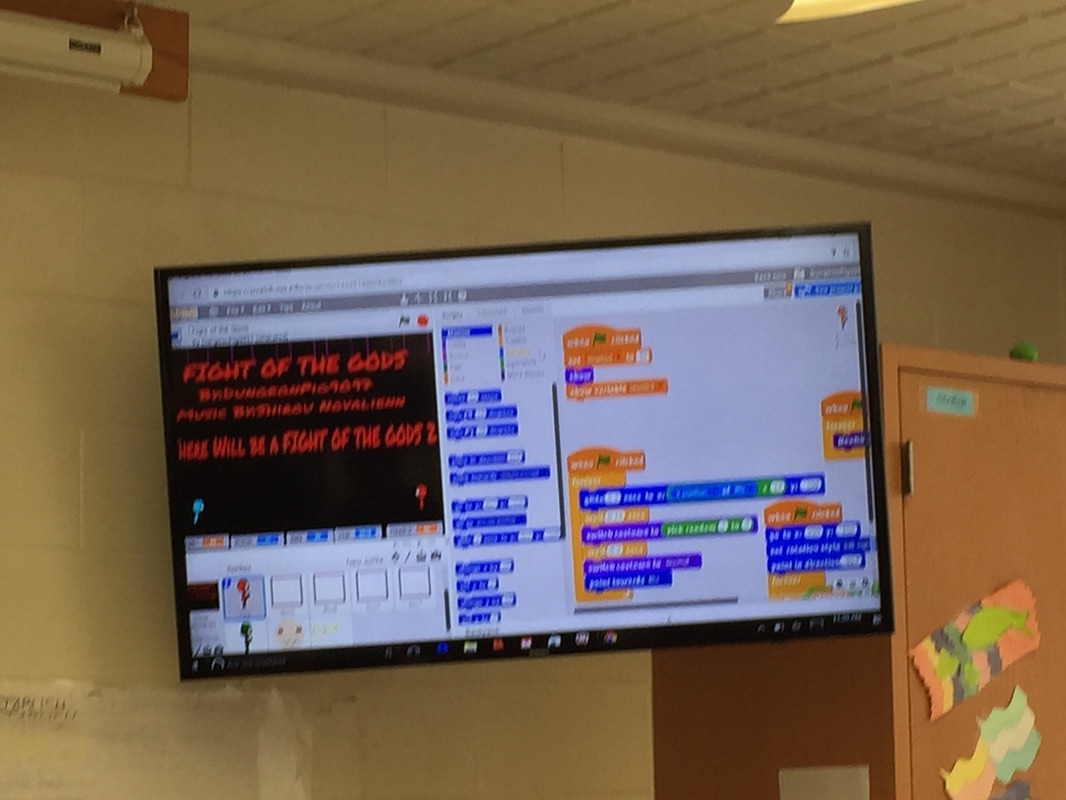
We spent a few weeks (from mid November up to Christmas break) to learn the simple yet super instructive Scratch computer programming language and created quite a few awesome video games, animation, and videos. I'm pretty impressed with how quickly the students caught on and how creative some of the games were in just a few short weeks! The students learned basic computer science concepts like creating sprites, how to instruct their characters to move, fly, jump, hop, run, disappear, and they learned about infinite loops, random number generators, score keeper, costume design, game theory, and much much more. For example, check out our most prolific Scratch progammer, Cameron's work by searching "pig9productions" on the Scratch website. I think you will be amazed!
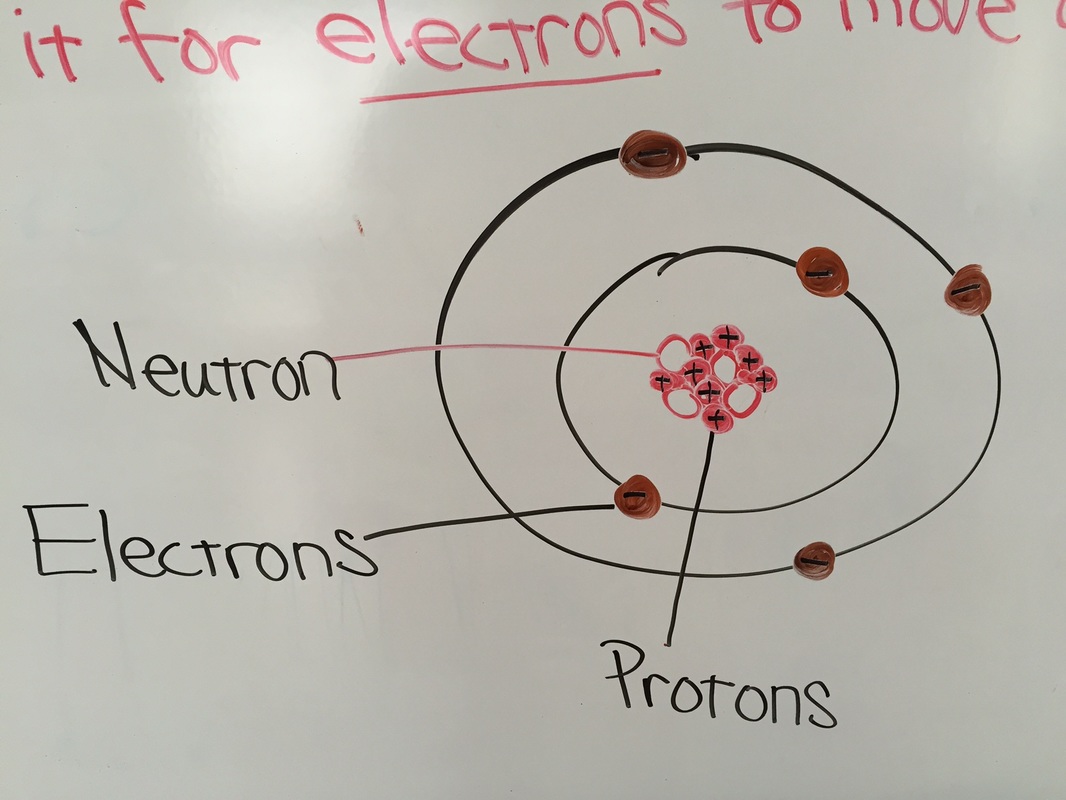
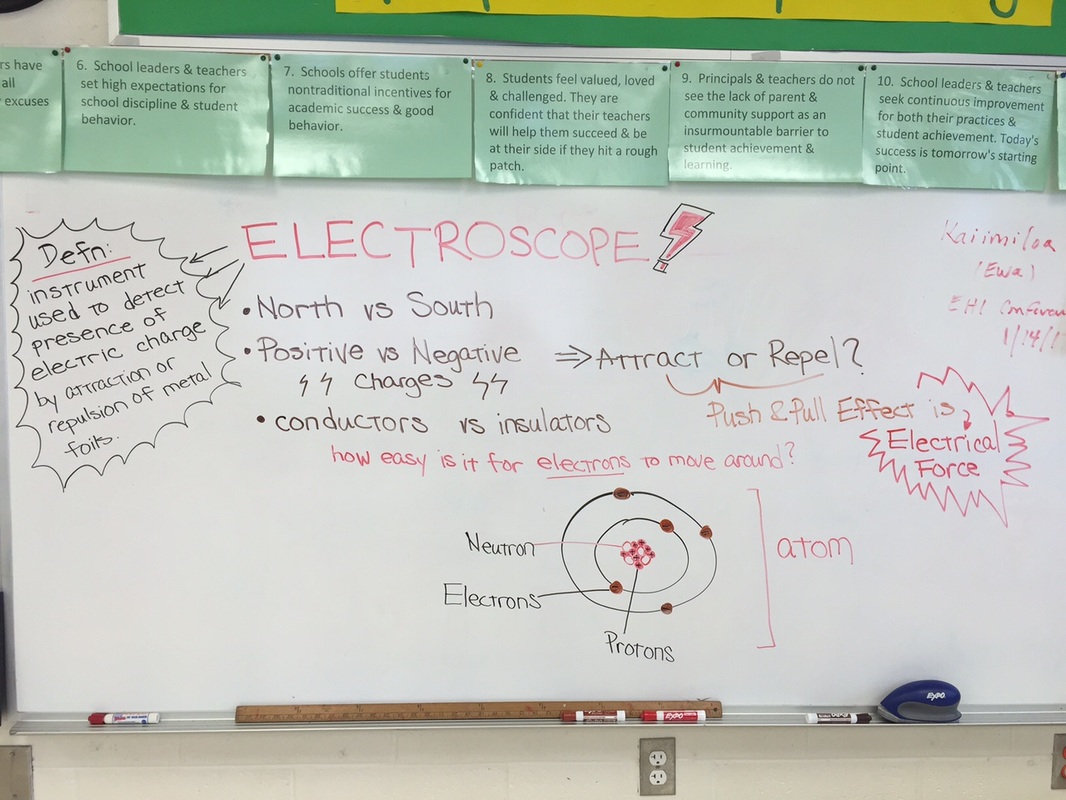
On 10/27 the 2nd and 3rd grade students learned about electrical force between two objects and built an electroscope! An electroscope is an instrument used to detect the presence of an electric charge by the amount of attraction or repulsion of metal foils. Every atom has negatively-charged electrons, positively-charged protons and neutral-charged neutrons. We also know that opposite charges attract and like charges repel or push away from each other. The amount of force depends on the amount of charge and the distance between the charges. This pushing or pulling force on the charge is called electrical force. And any two objects with different charges from each other have an electric force between them! The students had a lot of fun creating static electricity by rubbing a balloon on their hair and were amazed to see how the foil leaves react to this charge!
For those of you who are following our Biodome blog, here is a quick and fun update. All the biodomes are thriving and the students have had an awesome time making a home for baby caterpillars. Many of the caterpillars formed chrysalis and at least 5 butterflies have come out in the biodomes! We will continue to monitor these homes for little creatures and let the butterflies roam free when they come out. Stay tuned for more!
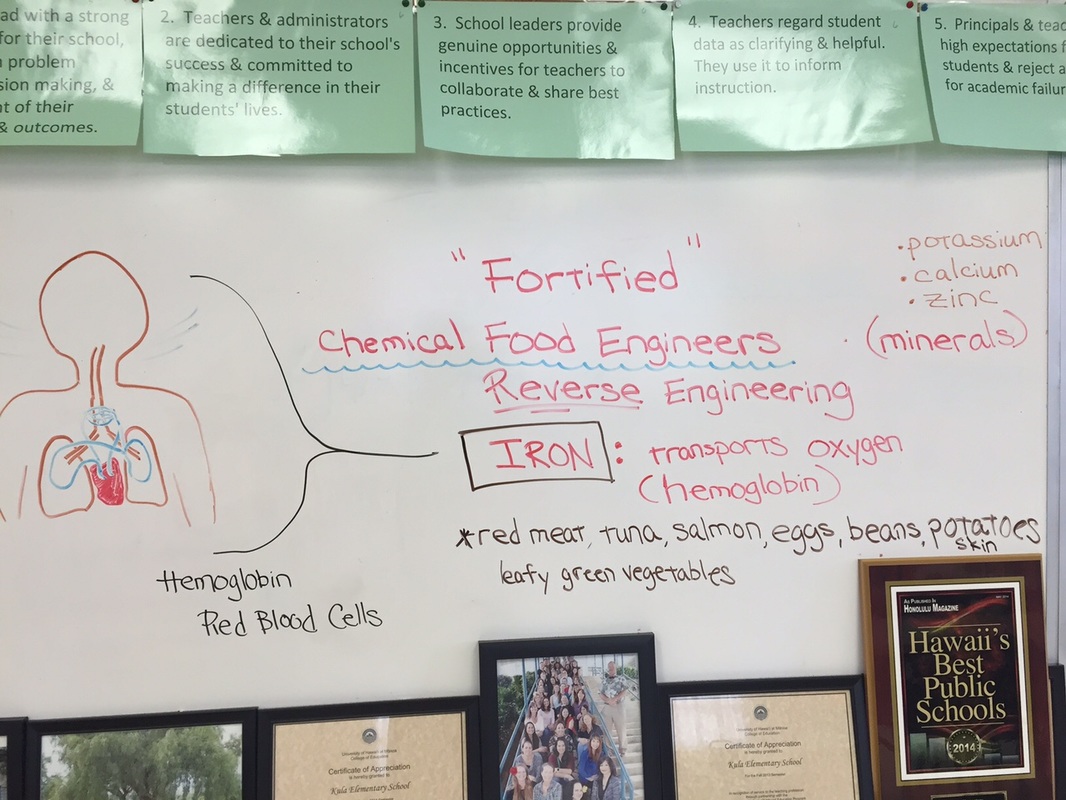
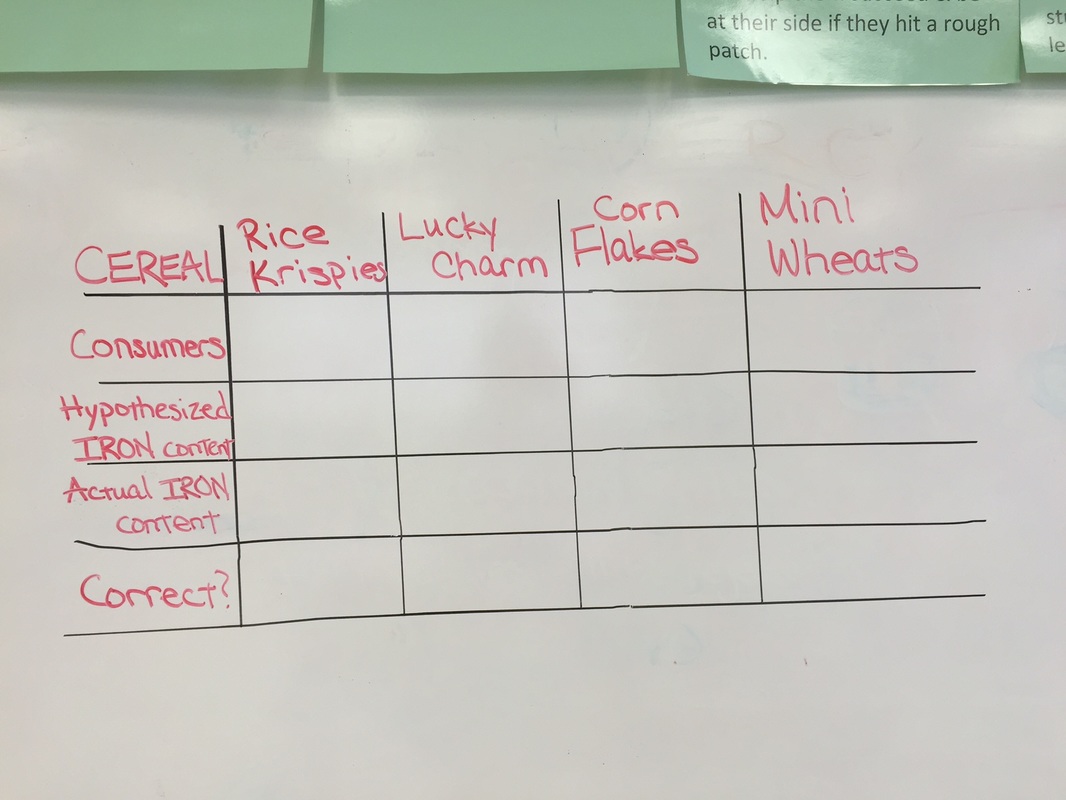
On 10/25, the 2nd and 3rd graders learned one of the roles of a Chemical Food Engineer and we got to experiment with some of our all-time favorite cereals. Chemical Food Engineers often fortify our foods like milk, juice, cereal etc. with additional vitamins and minerals to make sure that we all get our essential nutrients. Their role and their products are especially important when it comes to providing nutrients for people in areas where food is a scarce resource. The human body needs essential minerals like potassium, calcium, zinc, iron and many more to keep us healthy and functioning. For our class today, we are focusing on "iron." Iron is essential in forming hemoglobin which is part of our red blood cells. Hemoglobin transports oxygen in the blood from our lungs to the rest of the body. This is why iron is so important. In our experiment, we attempted to "extract" the iron from 4 popular cereals so that the students can actually see the iron with their naked eyes! To do this, we first had to crush up the cereal in a paper cup and then put it in water so the contents are freely flowing. Then, we used a super strong magnet (your average fridge magnet will not work for this!) and we slowly ran the magnet against the side of the cup and pulled the tiny slate specks of iron up to the surface. This was not an easy experiment but the students persevered and were quite shocked to find these little iron flakes in something they ate every morning. The winning cereal was Frosted Mini Wheats with and iron level of about 90% the daily recommended level!
On 10/20 the 3rd and 2nd graders embarked on a crystal growing adventure! With all the fun and festive Holidays just around the corner, I thought it would be a great idea to have the students learn how to grow their own crystals and explore the amazing chemistry behind it all. For our class, we decided to use Borax (the detergent booster) to grow our crystals but you can also make crystals out of more common household items like sugar and salt. We talked about solubility and saturation of the solution we are making with Borax and why heat is such an important element in making a solution that works. Not only does our Borax solution have to be super saturated, it has to be pure. Any impurities in the beaker may cause the crystals not to form correctly or it may cause a discoloration in the crystals. Crystals are formed through the process of nucleation. We explored the concept of a nucleus or a "seed" crystal - one in which other crystals start bonding and connecting to. We made hypothesis about the cooling temperature and environment and what would yield bigger, chunkier, fewer crystals vs small and numerous crystals. Check out the beautiful creations below - everyone took home an awesome crystal!
On Monday 10/24 ,the 4th and 5th graders were lucky to have a guest speaker. Dr. JD Armstrong from the Institute of Astronomy (just up the road from us on Kula Hwy.) is super involved in educating the kids in our community and was able to visit our STRETCH class and give a lesson on exoplanets.
Exoplanets are planets that do not orbit the sun and the students helped JD perform an experiment that demonstrates one of the methods used to "find" exoplanets. The method is called the transit method. Sometimes the planets orbiting other stars line up just right so that the planet passes between us and the star. When that happens the planet blocks a small portion of the light. We measure how bright the star is, and when we see the brightness drop down by a bit, we can tell that there is a planet there. To simulate this, JD used a big light bulb for the star, and a foam ball for the planet. The brightness of the bulb is measured using a photometer. The ball is moved a little way, in a circle around the bulb. The brightness of the bulb is measured and we repeat. JD had the students enter the photometer readings into a spreadsheet and help graph it out. The result is that there tends to be a big dip in the line graph (as a result of the change in the brightness of the light) when an exoplanet passes by. The students were super excited to participate and help the demo. They also asked many questions about dwarf planets as well as the middle/high school astronomy program that JD heads up. Thanks again Dr. JD Armstrong and be sure to catch the next open house at the Institute of Astronomy! You get to use super powerful telescopes to see the moon and planets - don't miss out! So far in STRETCH, we've done experiments in chemistry and physics. During the week of October 3, we switched gears to explore biology and started a series on biodomes! Now if you are as much into catching bugs as I am, this series is for you! A biodome is a self-sufficient ecosystem that is build to replicate one of the biomes (large outdoor areas on the earth's surface with animals and plants which have adapted to the natural environment - climate, geology, soil, vegetation, etc.). It's great for us scientists because it's a controlled space - one in which we can gleam a lot from. We reviewed the different biomes with the students and for the sake of our experiment, we kept it to these 5 general groups: tundra, forest, grassland, desert, and aquatic. We also discussed producers, consumers, and decomposers in an ecosystem.
This project requires many sessions as we need to first make a "home" for our producers - our food source! Check out the diagram below for how we were able to fashion a simple and self-sufficient biodome just out of plastic bottles, string, and some plotting soil. Each team tailored their biodome a bit depending on one of the 5 biomes they were assigned to replicate. Each group also presented their plans for their biodomes in front of the class. In the interest of time and factoring in the weather, we decided on planting green beans as our food source. I'm super excited to report that after just one week of fall break (no watering and completely self-sustained) our biodomes are working successfully and there are huge, healthy stalks of bean plants in every biodome! Now the next phase is to find some creepy crawling creatures around the school yard and put them into the biodome as our consumers. The last phase would be to find the slimy wormy decomposers - snails, slugs, and worms! |
AuthorsMrs. Shim and her STRETCH students Archives
May 2015
Categories |










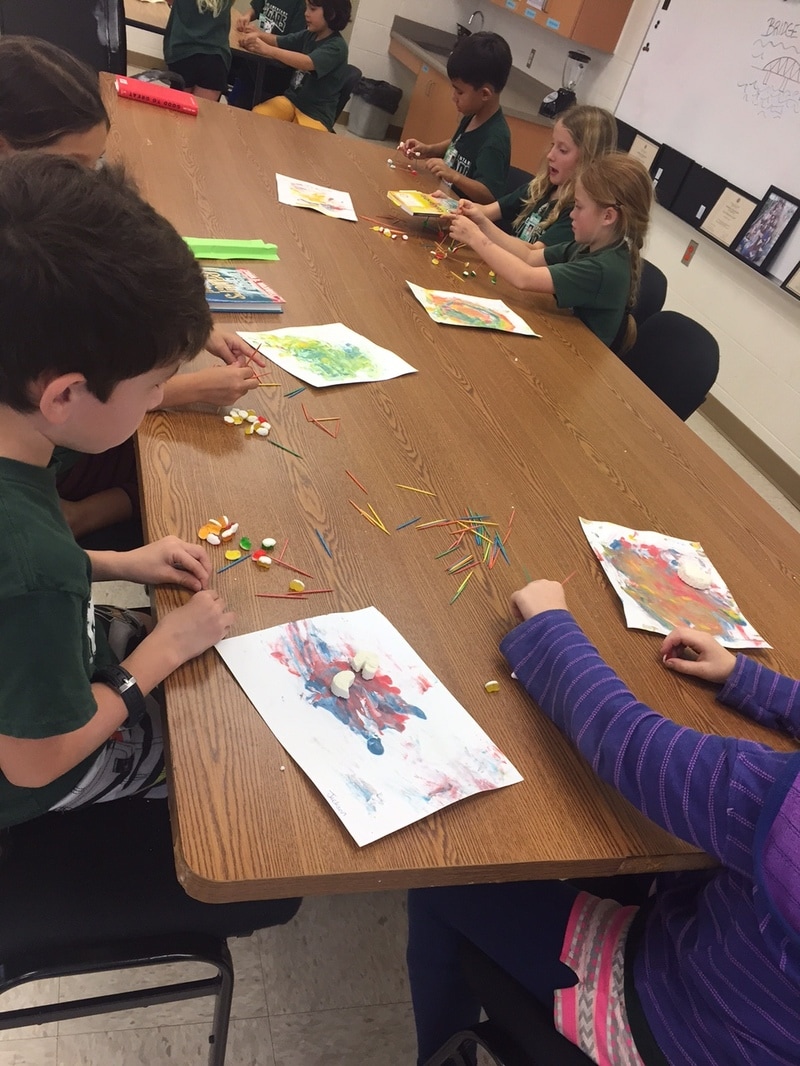





























































































 RSS Feed
RSS Feed
